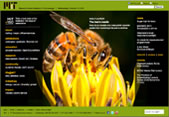
Today’s spotlight features an illustration, by Wikimedia Commons user Dcrjsr and edited by MIT News, of a 4-stranded antiparallel beta sheet fragment from a crystal structure of the enzyme catalase.
Proteins can fold in different ways depending on their environment. These different configurations change the function of the protein; misfolding is frequently associated with diseases such as Alzheimer’s and Parkinson’s.
Until now, it has been difficult to fully characterize the different structures that proteins can take on in their natural environments. However, using a new technique known as sensitivity-enhanced nuclear magnetic resonance, MIT researchers have shown that they can analyze the structure that a yeast protein forms as it interacts with other proteins in a cell.
Read full article.
Proteins can fold in different ways depending on their environment. These different configurations change the function of the protein; misfolding is frequently associated with diseases such as Alzheimer’s and Parkinson’s.
Until now, it has been difficult to fully characterize the different structures that proteins can take on in their natural environments. However, using a new technique known as sensitivity-enhanced nuclear magnetic resonance, MIT researchers have shown that they can analyze the structure that a yeast protein forms as it interacts with other proteins in a cell.
Read full article.