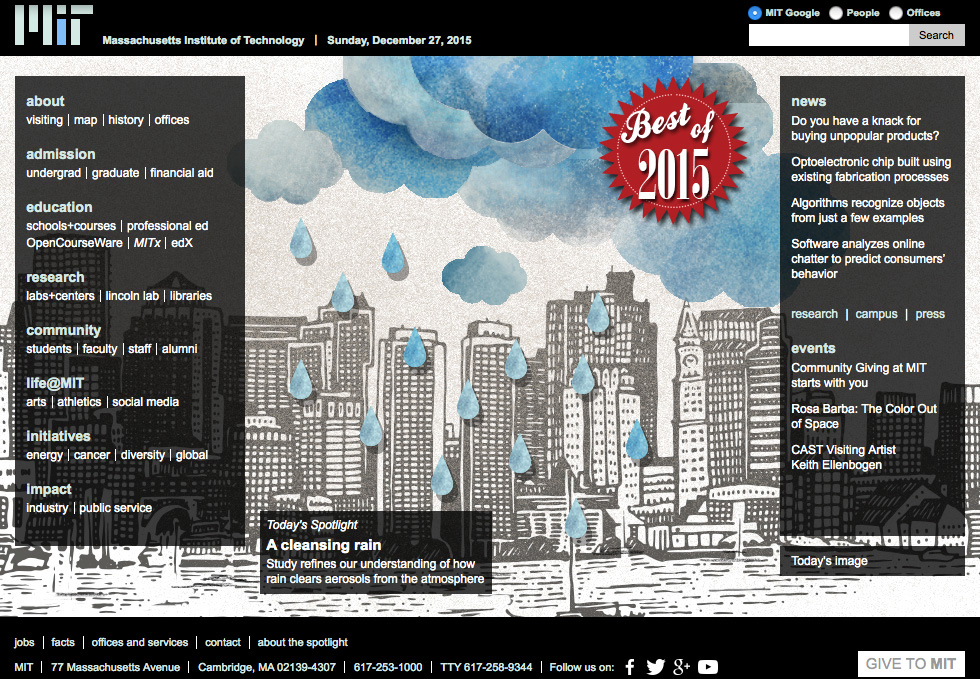
Today’s Spotlight features an illustration by Christine Daniloff/MIT.
As a raindrop falls through the atmosphere, it can attract tens to hundreds of tiny aerosol particles to its surface before hitting the ground. The process by which droplets and aerosols attract is coagulation, a natural phenomenon that can act to clear the air of pollutants like soot, sulfates, and organic particles.
Atmospheric chemists at MIT have now determined just how effective rain is in cleaning the atmosphere. Given the altitude of a cloud, the size of its droplets, and the diameter and concentration of aerosols, the team can predict the likelihood that a raindrop will sweep a particle out of the atmosphere.
Read full article.
As a raindrop falls through the atmosphere, it can attract tens to hundreds of tiny aerosol particles to its surface before hitting the ground. The process by which droplets and aerosols attract is coagulation, a natural phenomenon that can act to clear the air of pollutants like soot, sulfates, and organic particles.
Atmospheric chemists at MIT have now determined just how effective rain is in cleaning the atmosphere. Given the altitude of a cloud, the size of its droplets, and the diameter and concentration of aerosols, the team can predict the likelihood that a raindrop will sweep a particle out of the atmosphere.
Read full article.